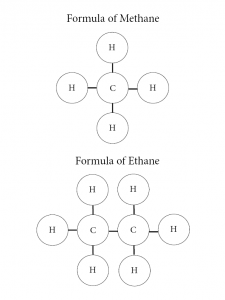
It’s never been better to study something new, especially when it comes to chemistry. Carbon is basically the main focus of this round, but what is carbon? What makes Carbon so special that we focus on only carbon the whole seven weeks? Well, we didn’t actually spend the whole 7 weeks, because we only study stem every other week, which means we only studied about three weeks or so. Back to the topic, Carbon is everywhere, because 0.04% of the atmosphere is made out of Carbon. Carbon is known as the “element of life”, because all living things contain carbon in them. Besides just carbon, we studied hydrocarbons and carbon dioxide. Hydrocarbon is a compound combination of hydrogen and carbon only. Carbon needs to bond with four other elements around it to be a complete hydrocarbon. There are different prefix for different hydrocarbon, such as:
Meth: 4 hydrogens and 1 carbon
Eth: 6 hydrogens and 2 carbon
Prop: 8 hydrogens and 3 carbon
But: 10 hydrogens and 4 carbon
Pent: 12 hydrogens and 5 carbon
Hex: 14 hydrogens and 6 carbon
Hept: 16 hydrogens and 7 carbon
Oct: 18 hydrogens and 8 carbon
Non: 20 hydrogens and 9 carbon
Dec: 22 hydrogens and 10 carbon
Below shows some examples of hydrocarbon.
Afterward we did a black snake experiment and wrote a lab report about the experiment.